AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Cdc up to date vaccine11/27/2023  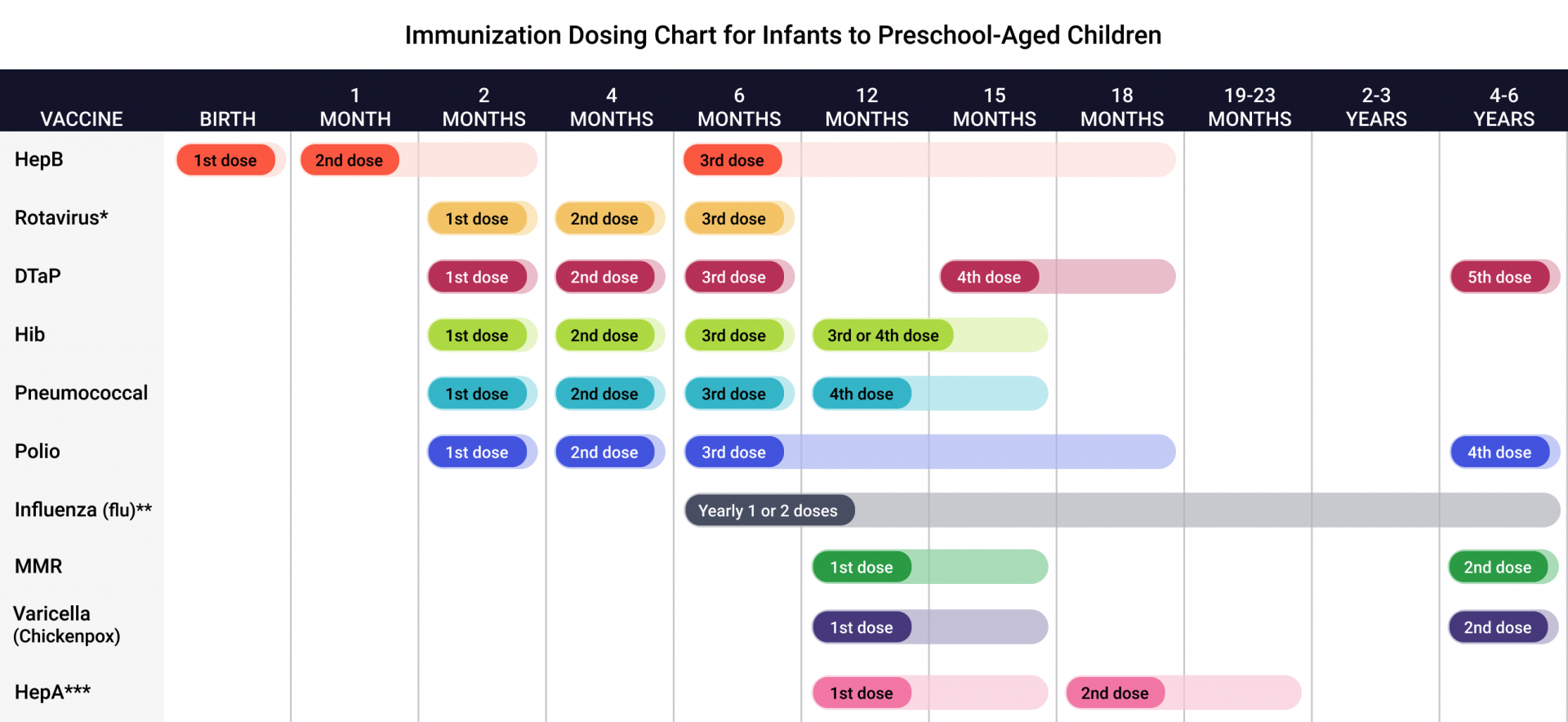
Although bivalent booster vaccination coverage among adults differed by factors such as income, health insurance status, and social vulnerability index (SVI), these factors were not associated with differences in reluctance to seek booster vaccination. Among adolescents with parents who were open to getting a booster vaccination for their child, 32.4% had not received a provider recommendation for any COVID-19 vaccination, and 11.8% had parents who reported safety concerns. Among adults who were open to receiving booster vaccination, 58.9% reported not having received a provider recommendation for booster vaccination, 16.9% had safety concerns, and 4.4% reported difficulty getting a booster vaccine. Bivalent booster coverage was lower among non-Hispanic Black or African American (Black) and Hispanic or Latino (Hispanic) adolescents and adults compared with non-Hispanic White (White) adolescents and adults. Adolescents and adults in rural areas had a much lower primary series completion rate and up-to-date vaccination coverage. Based on data collected during October 30–December 31, 2022, from the National Immunization Survey–Adult COVID Module (NIS-ACM) ( 4), 27.1% of adults who had completed a COVID-19 primary series had received a bivalent booster, 39.4% had not yet received a bivalent booster but were open to receiving booster vaccination, 12.4% had not yet received a bivalent booster and were unsure about getting a booster vaccination, and 21.1% were reluctant to receive a booster. 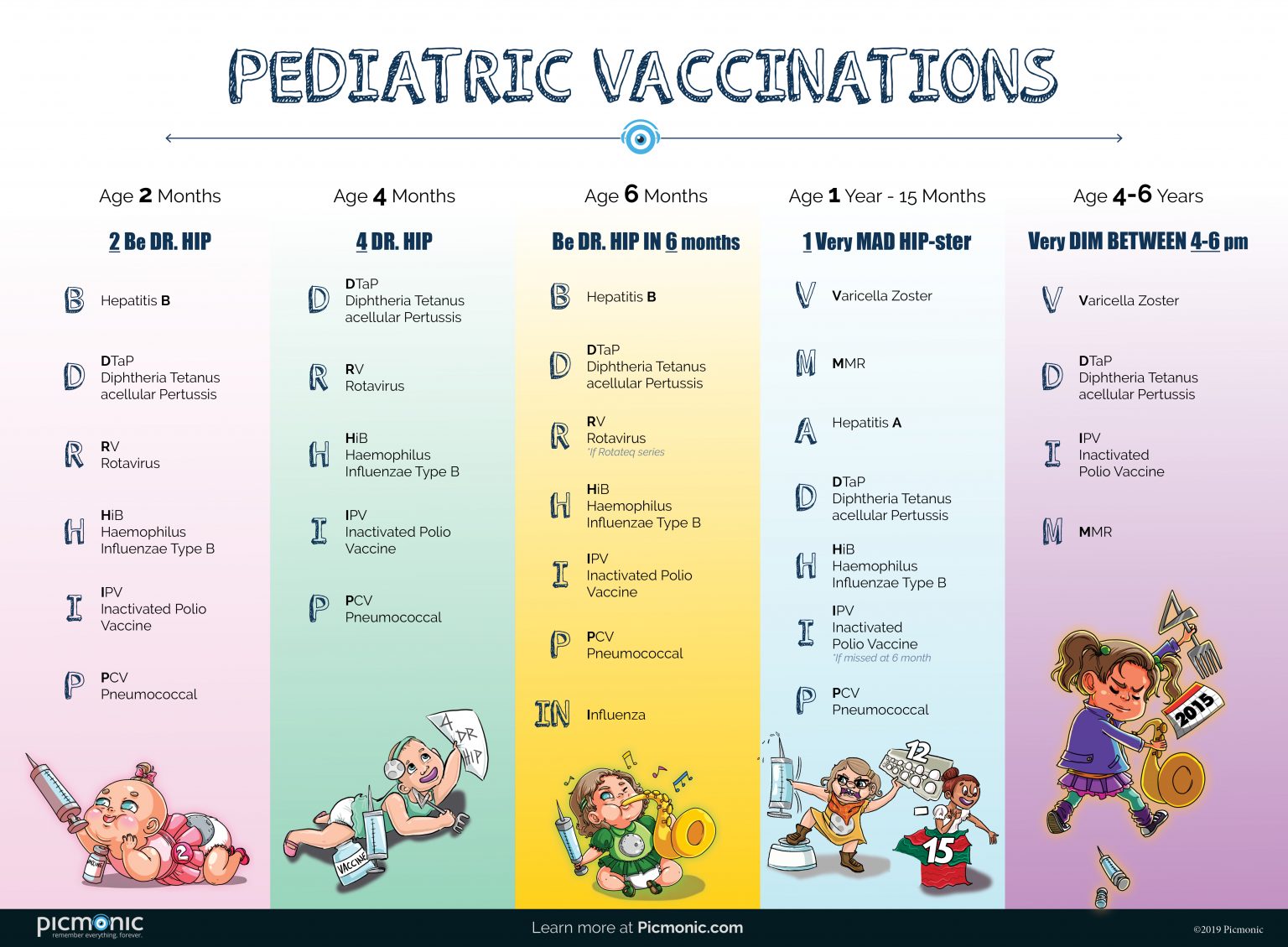
Based on data collected during October 30–December 31, 2022, from the National Immunization Survey–Child COVID Module (NIS-CCM) ( 4), among all adolescents aged 12–17 years who completed a primary series, 18.5% had received a bivalent booster dose, 52.0% had not yet received a bivalent booster but had parents open to booster vaccination for their child, 15.1% had not received a bivalent booster and had parents who were unsure about getting a booster vaccination for their child, and 14.4% had parents who were reluctant to seek booster vaccination for their child. The bivalent booster is formulated to protect against the Omicron BA.4 and BA.5 subvariants of SARS-CoV-2 as well as the original (ancestral) strain ( 3). CDC recommended an updated (bivalent) booster for adolescents aged 12–17 years and adults aged ≥18 years on Septem( 3). Learn more about adverse events, including reports of death, after COVID-19 vaccination.COVID-19 vaccine booster doses are safe and maintain protection after receipt of a primary vaccination series and reduce the risk for serious COVID-19–related outcomes, including emergency department visits, hospitalization, and death ( 1, 2). CDC and FDA review reports of death following COVID-19 vaccination and update information as it becomes available. Reports of adverse events to VAERS following vaccination, including deaths, do not necessarily mean that a vaccine caused a health problem. FDA requires healthcare providers to report any death after COVID-19 vaccination to the Vaccine Adverse Event Reporting System (VAERS), even if it’s unclear whether the vaccine was the cause. Reports of death after COVID-19 vaccination are rare. Learn more about COVID-19 vaccines and adverse events, including GBS. Guillain-Barré Syndrome (GBS) is a rare disorder where the body’s immune system damages nerve cells, causing muscle weakness and sometimes paralysis. Learn more about COVID-19 vaccines and adverse events, including myocarditis and pericarditis. Myocarditis and pericarditis after COVID-19 vaccination are rare. Myocarditis is inflammation of the heart muscle, and pericarditis is inflammation of the outer lining of the heart. Learn more about COVID-19 vaccines and adverse events, including TTS. Thrombosis with thrombocytopenia syndrome (TTS) is a rare but serious adverse event that causes blood clots or issues with clotting. Thrombosis with Thrombocytopenia Syndrome (TTS) Learn more about COVID-19 vaccines and allergic reactions, including anaphylaxis. Anaphylaxis after COVID-19 vaccination is rare. Anaphylaxis is a severe type of allergic reaction with symptoms such as hives, difficulty breathing, low blood pressure, or significant swelling of the tongue or lips. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
